Pill With Imprint 717
This page shows all pills in our database with the imprint code 717. Imprint codes are stamped or printed on pill tablets and capsules and are used to uniquely identify medications. Data is sourced from the FDA NDC Directory and DailyMed.
Found 9 pills with imprint 717
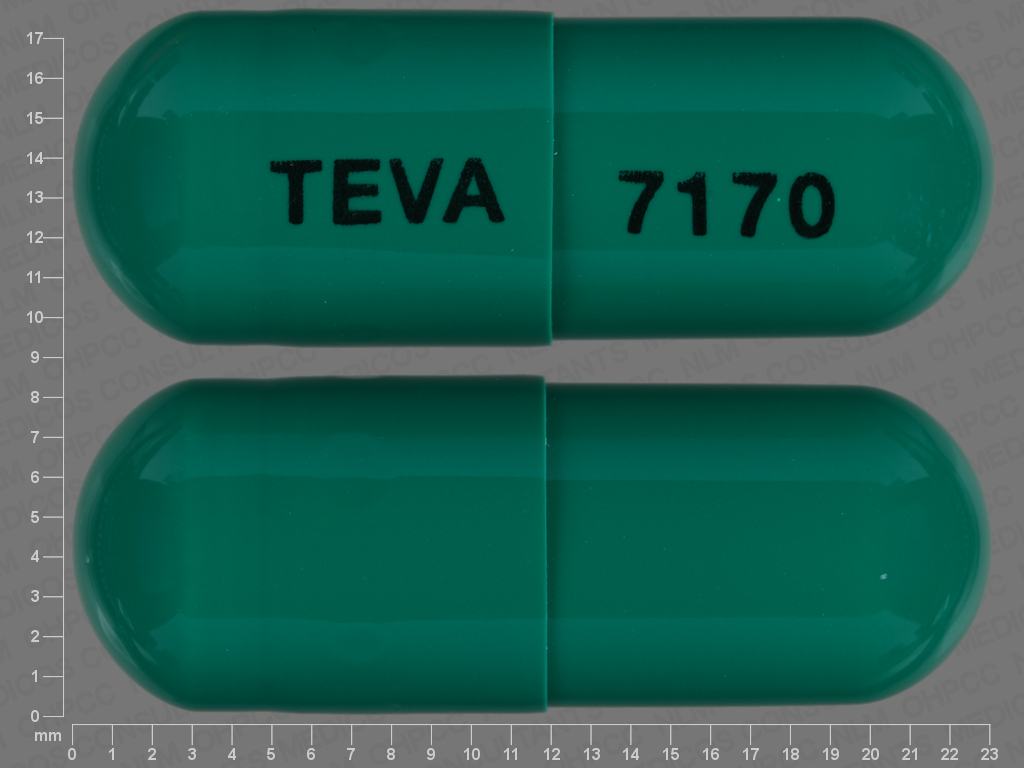
celecoxib
400 MG
NDC: 00093-7170-06
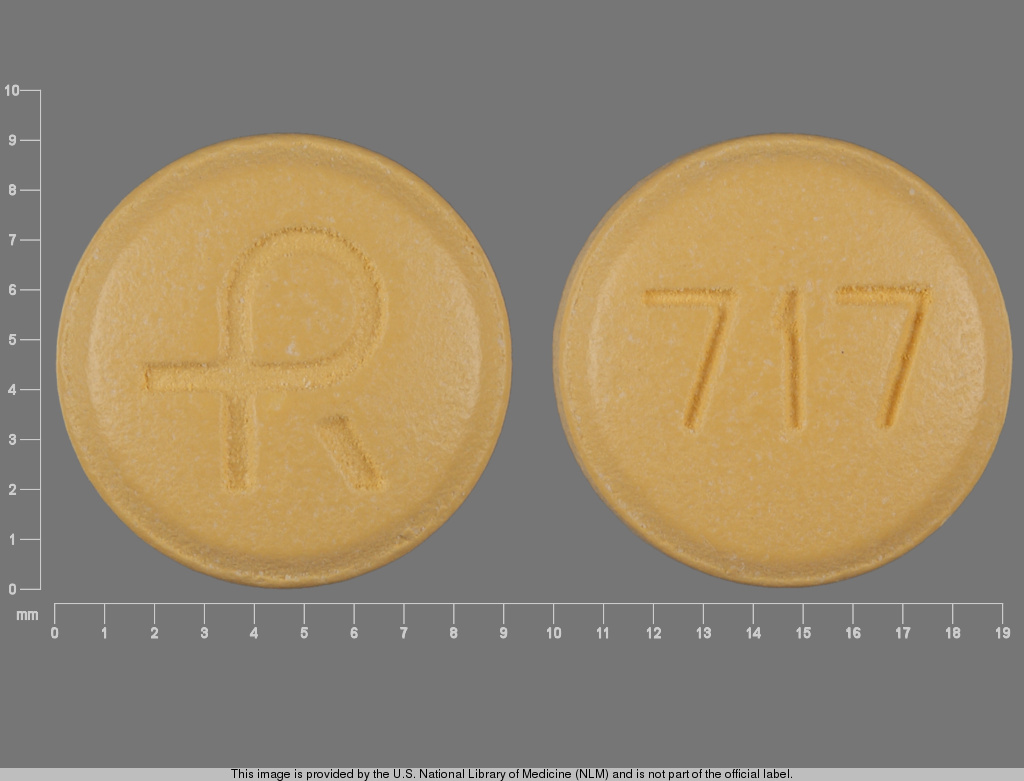
Diclofenac Sodium
DICLOFENAC SODIUM 100 mg;

Etodolac
ETODOLAC 500 mg;

Hydrochlorothiazide/Losartan Potassium
12.5 MG/50 MG
NDC: 00006-0717-31

HYZAAR
losartan potassium 50 mg;hydrochlorothiazide 12.5 mg;

losartan potassium and hydrochlorothiazide
losartan potassium 50 mg;hydrochlorothiazide 12.5 mg;

Nefazodone Hydrochloride
NEFAZODONE HYDROCHLORIDE 50 mg;

Triazolam
TRIAZOLAM 0.125 mg;

Triazolam
0.125 MG
NDC: 59762-3717-04
What is an imprint code?
An imprint code is a combination of letters, numbers, or symbols that appears on a pill, tablet, or capsule. The FDA requires all approved drug products to have a unique imprint so that they can be identified in case of accidental ingestion or medication confusion. The imprint 717 is the code found on the pill you are looking up.
⚠️ Disclaimer: Pill identification is for educational purposes only. Always confirm identification with a licensed pharmacist. Read full disclaimer.