Pill With Imprint B
This page shows all pills in our database with the imprint code B. Imprint codes are stamped or printed on pill tablets and capsules and are used to uniquely identify medications. Data is sourced from the FDA NDC Directory and DailyMed.
Found 46 pills with imprint B

Microencapsulated Potassium Chloride
20 MEQ
NDC: 62037-0999-10
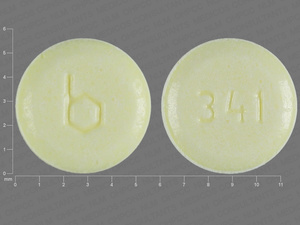
{12 (Ethinyl Estradiol/Norethindrone/9 (Ethinyl Estradiol/Norethindrone/7 (Inert Ingredients
0.035 MG/0.5 MG/0.035 MG/1 MG/1 MG
NDC: 00555-9066-67

Mimvey
ESTRADIOL 1 mg;NORETHINDRONE ACETATE 0.5 mg;

Mimvey Lo
ESTRADIOL 0.5 mg;NORETHINDRONE ACETATE 0.1 mg;

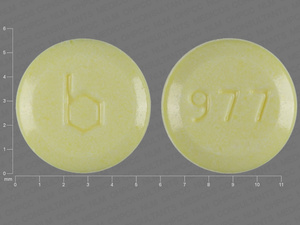
{21 (Ethinyl Estradiol/norethindrone acetate/7 (Ferrous fumarate
0.02 MG/1 MG/75 MG
NDC: 00555-9026-58
{21 (Ethinyl Estradiol/norethindrone acetate/7 (Ferrous fumarate
0.02 MG/1 MG/75 MG
NDC: 00555-9026-58
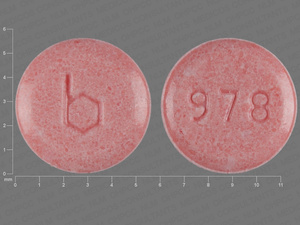
{21 (Ethinyl Estradiol/norethindrone acetate/7 (Ferrous fumarate
0.03 MG/1.5 MG/75 MG
NDC: 00555-9028-58

{5 (Ethinyl Estradiol/norethindrone acetate/7 (Ethinyl Estradiol/norethindrone acetate/9 (Ethinyl Estradiol/norethindrone acetate/7 (Ferrous fumarate
0.02 MG/1 MG/0.03 MG/1 MG/0.035 MG/1 MG/75 MG
NDC: 00555-9032-70

{7 (Desogestrel/Ethinyl Estradiol/7 (Desogestrel/Ethinyl Estradiol/7 (Desogestrel/Ethinyl Estradiol/7 (Inert Ingredients
0.1 MG/0.025 MG/0.125 MG/0.025 MG/0.15 MG/0.025 MG/1 MG
NDC: 00555-9051-67

{7 (Ethinyl Estradiol/84 (Ethinyl Estradiol/Levonorgestrel
0.01 MG/0.03 MG/0.15 MG
NDC: 00093-3134-82

{7 (Ethinyl Estradiol/84 (Ethinyl Estradiol/Levonorgestrel
0.01 MG/0.02 MG/0.1 MG
NDC: 51285-0092-87

{7 (Ethinyl Estradiol/Norethindrone/7 (Ethinyl Estradiol/Norethindrone/7 (Ethinyl Estradiol/Norethindrone/7 (Inert Ingredients
0.035 MG/0.5 MG/0.035 MG/0.75 MG/0.035 MG/1 MG/1 MG
NDC: 00555-9012-58

{7 (Ethinyl Estradiol/norgestimate/7 (Ethinyl Estradiol/norgestimate/7 (Ethinyl Estradiol/norgestimate/7 (Inert Ingredients
0.025 MG/0.18 MG/0.025 MG/0.215 MG/0.025 MG/0.25 MG/1 MG
NDC: 00093-2140-62

{7 (Ethinyl Estradiol/norgestimate/7 (Ethinyl Estradiol/norgestimate/7 (Ethinyl Estradiol/norgestimate/7 (Inert Ingredients
0.025 MG/0.18 MG/0.025 MG/0.215 MG/0.025 MG/0.25 MG/1 MG
NDC: 00093-2140-62

{7 (Ethinyl Estradiol/norgestimate/7 (Ethinyl Estradiol/norgestimate/7 (Ethinyl Estradiol/norgestimate/7 (Inert Ingredients
0.035 MG/0.18 MG/0.035 MG/0.215 MG/0.035 MG/0.25 MG/1 MG
NDC: 00555-9018-58

{84 (Ethinyl Estradiol/Levonorgestrel/7 (Inert Ingredients
0.03 MG/0.15 MG/1 MG
NDC: 00555-9123-66

Amoxicillin
775 MG
NDC: 68453-0142-03

Ascomp with Codeine
Butalbital 50 mg;Aspirin 325 mg;Caffeine 40 mg;Codeine Phosphate 30 mg;
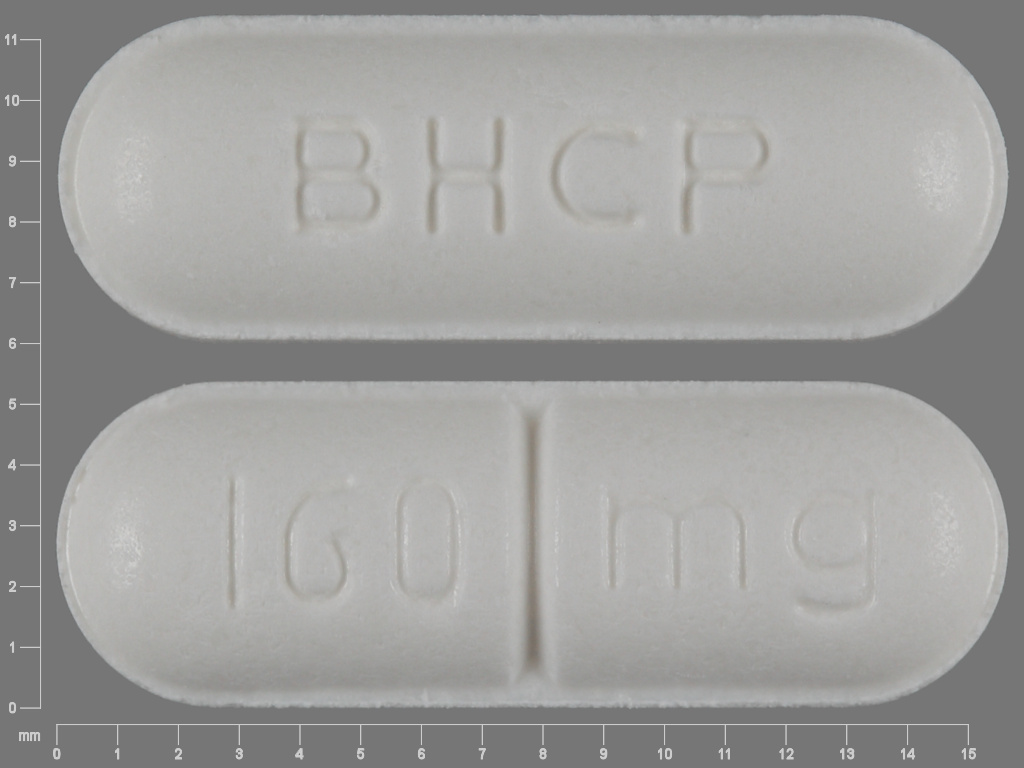
Betapace AF
SOTALOL HYDROCHLORIDE 160 mg;
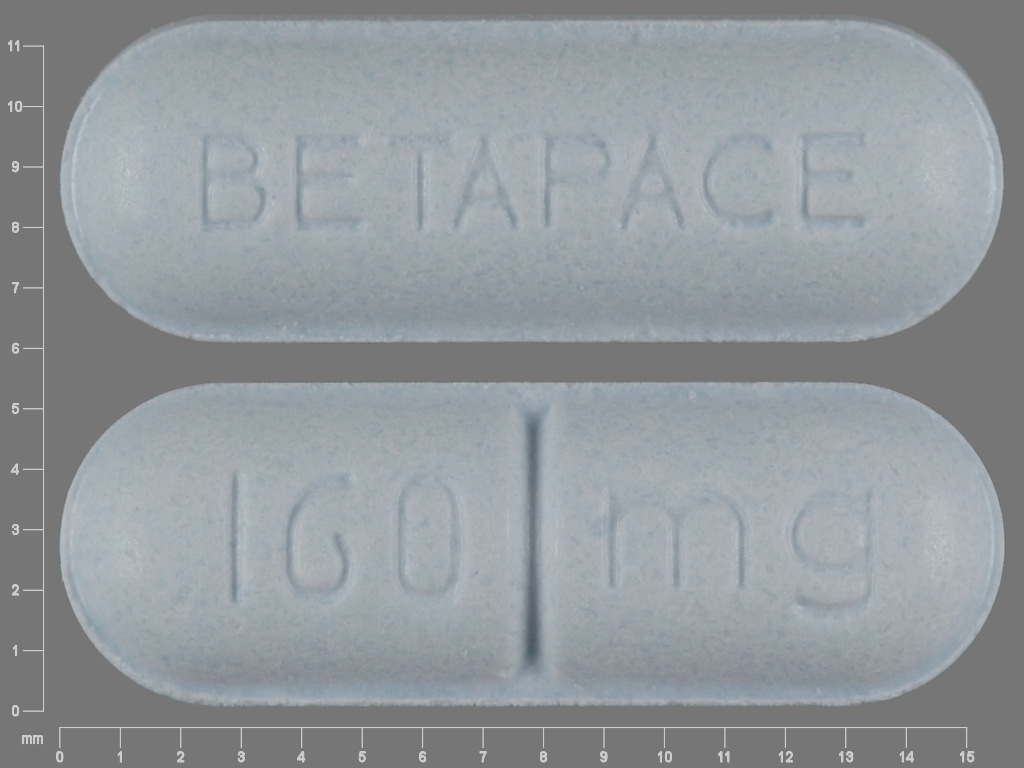
Betapace
SOTALOL HYDROCHLORIDE 160 mg;
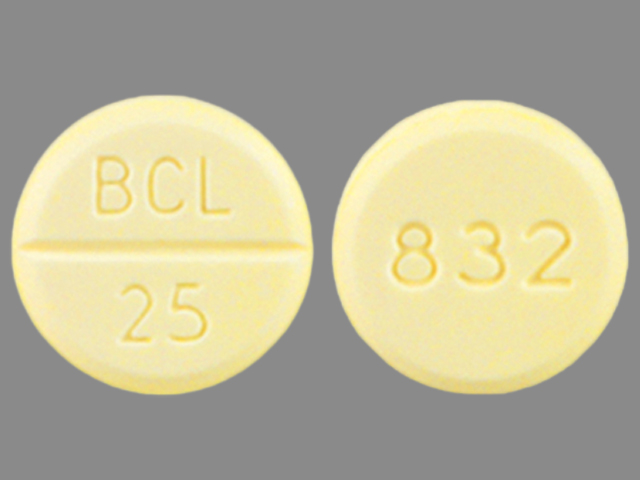
Bethanechol Chloride
Bethanechol Chloride 25 mg;
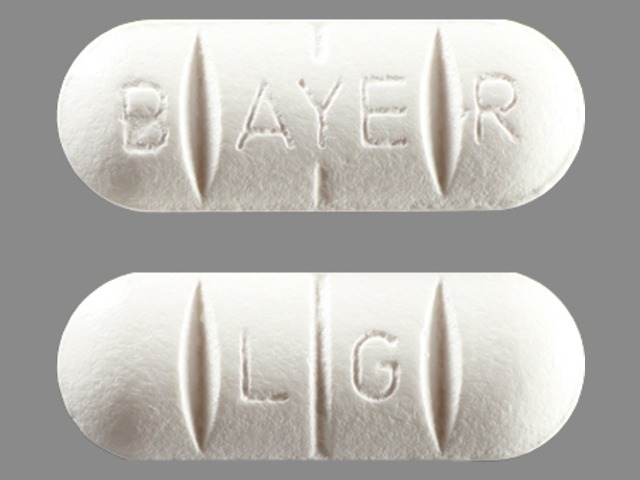
Biltricide
PRAZIQUANTEL 600 mg;

Buprenorphine
2 MG
NDC: 00093-5378-56
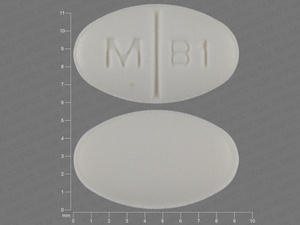
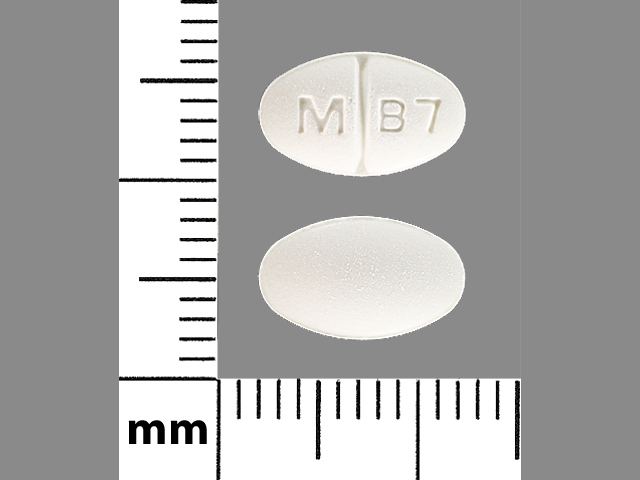
buspirone hydrochloride
5 MG
NDC: 00378-1140-01
Buspirone Hydrochloride
BUSPIRONE HYDROCHLORIDE 7.5 mg;
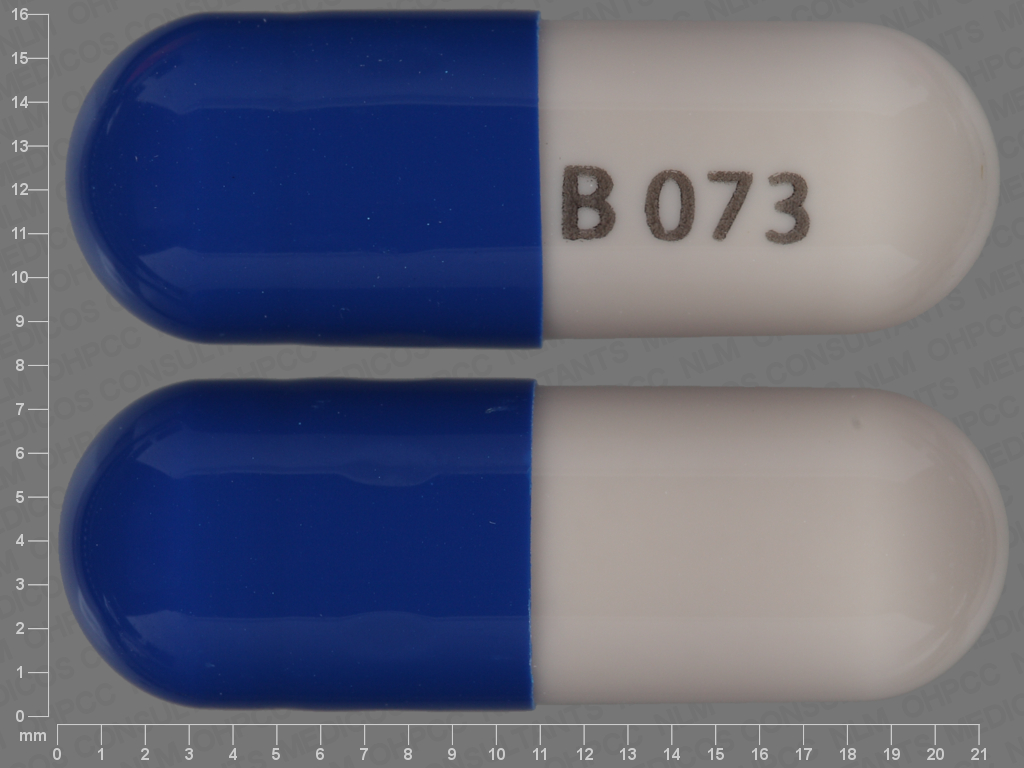
Butalbital, Acetaminophen, Caffeine and Codeine Phosphate
Butalbital 50 mg;Acetaminophen 325 mg;Caffeine 40 mg;Codeine Phosphate 30 mg;
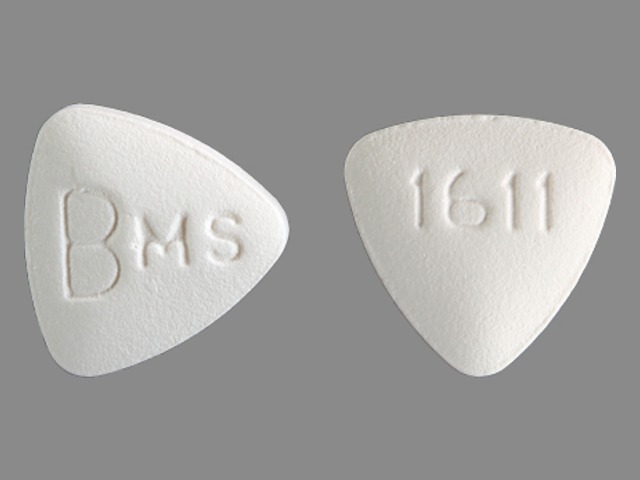
BARACLUDE
ENTECAVIR 0.5 mg;
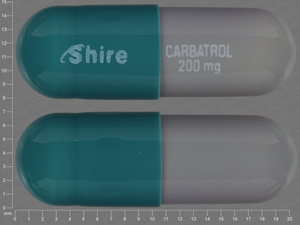
Carbamazepine
200 MG
NDC: 54092-0172-12

Catapres
clonidine hydrochloride 0.100 mg;
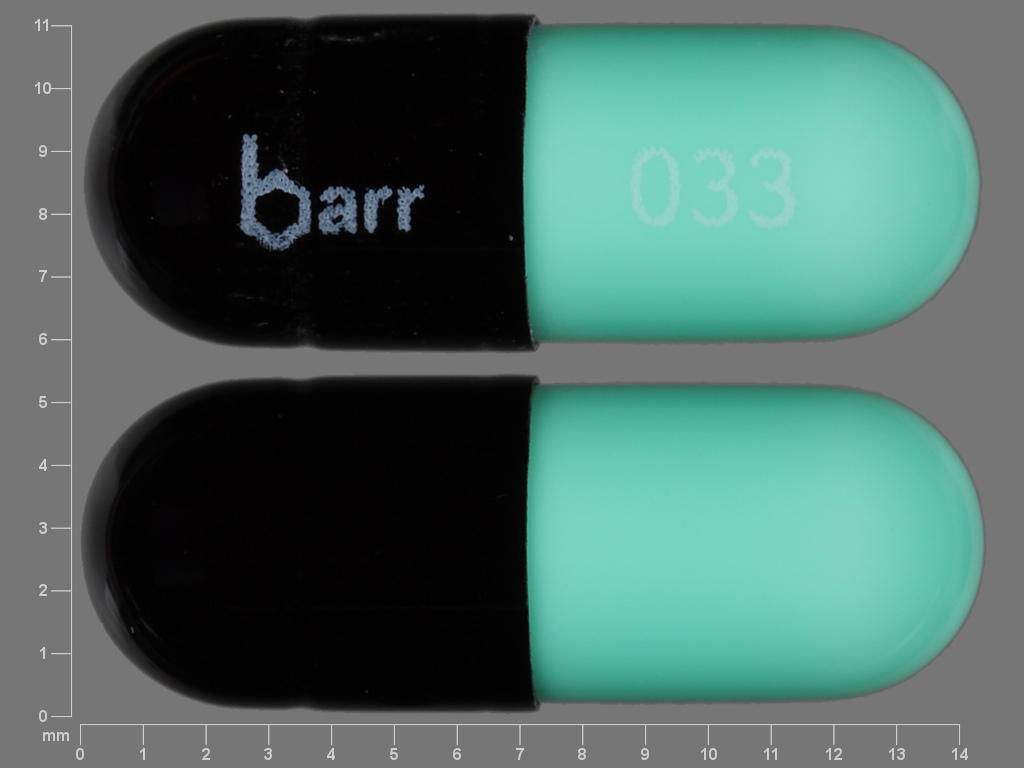
Chlordiazepoxide Hydrochloride
CHLORDIAZEPOXIDE HYDROCHLORIDE 10 mg;
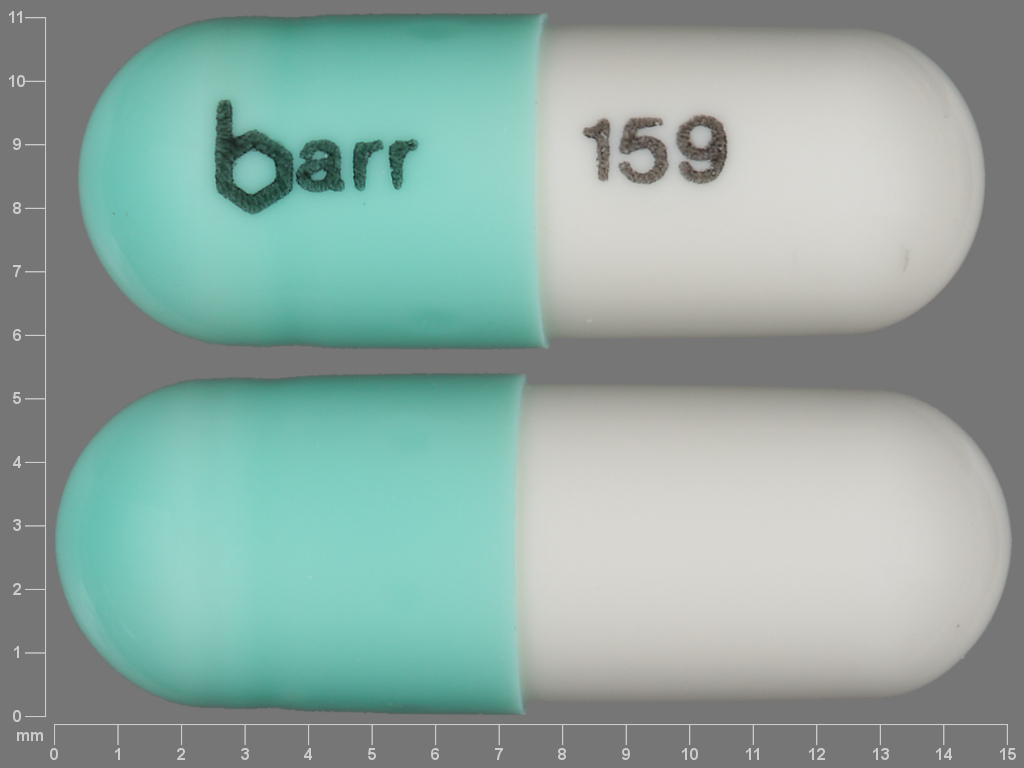
Chlordiazepoxide Hydrochloride
CHLORDIAZEPOXIDE HYDROCHLORIDE 25 mg;
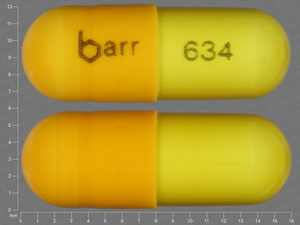
Danazol
100 MG
NDC: 00555-0634-02

Dextroamphetamine Saccharate, Amphetamine Aspartate, Dextroamphetamine Sulfate and Amphetamine Sulfate
DEXTROAMPHETAMINE SACCHARATE 1.875 mg;AMPHETAMINE ASPARTATE MONOHYDRATE 1.875 mg;DEXTROAMPHETAMINE SULFATE 1.875 mg;AMPHETAMINE SULFATE 1.875 mg;
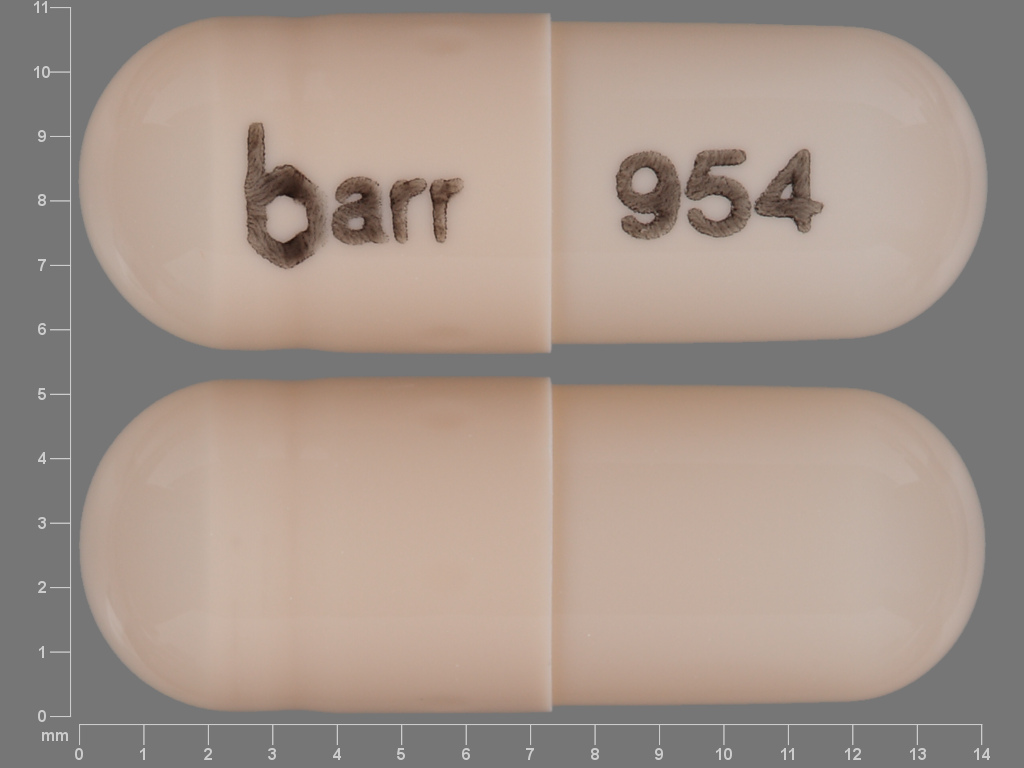
Dextroamphetamine Sulfate
DEXTROAMPHETAMINE SULFATE 5 mg;

Didanosine
DIDANOSINE 200 mg;
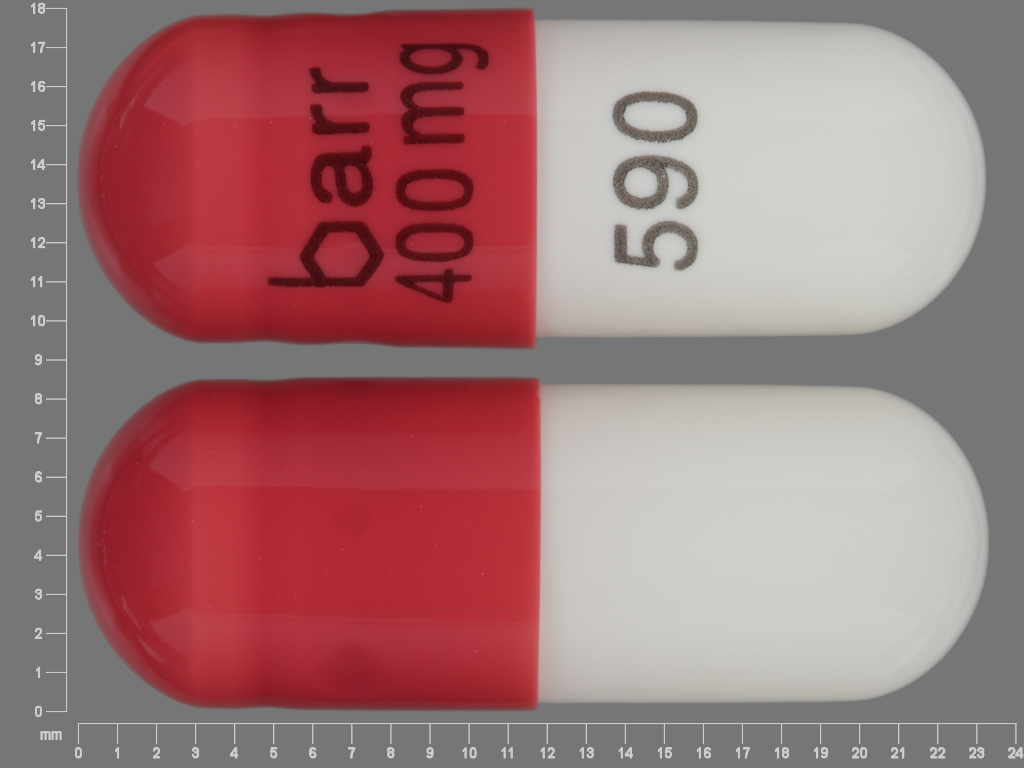
Didanosine
DIDANOSINE 400 mg;

Diltiazem Hydrochloride
180 MG
NDC: 00074-3061-90

Diltiazem Hydrochloride
Diltiazem Hydrochloride 420 mg;

Diltiazem Hydrochloride
Diltiazem Hydrochloride 240 mg;

Diphenhydramine Hydrochloride
DIPHENHYDRAMINE HYDROCHLORIDE 50 mg

Acetazolamide
ACETAZOLAMIDE 500 mg;
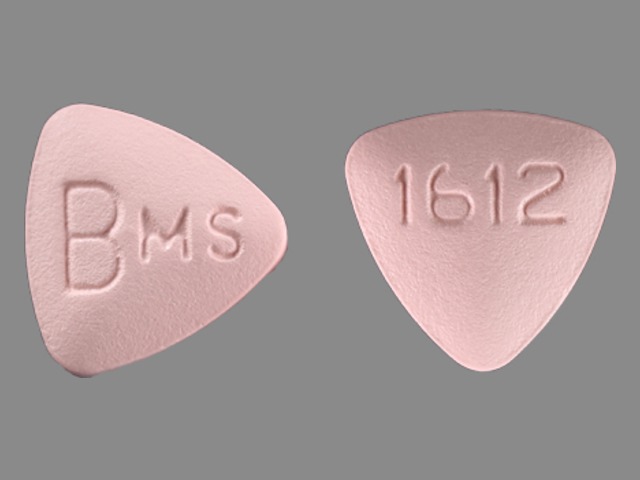
BARACLUDE
ENTECAVIR 1.0 mg;
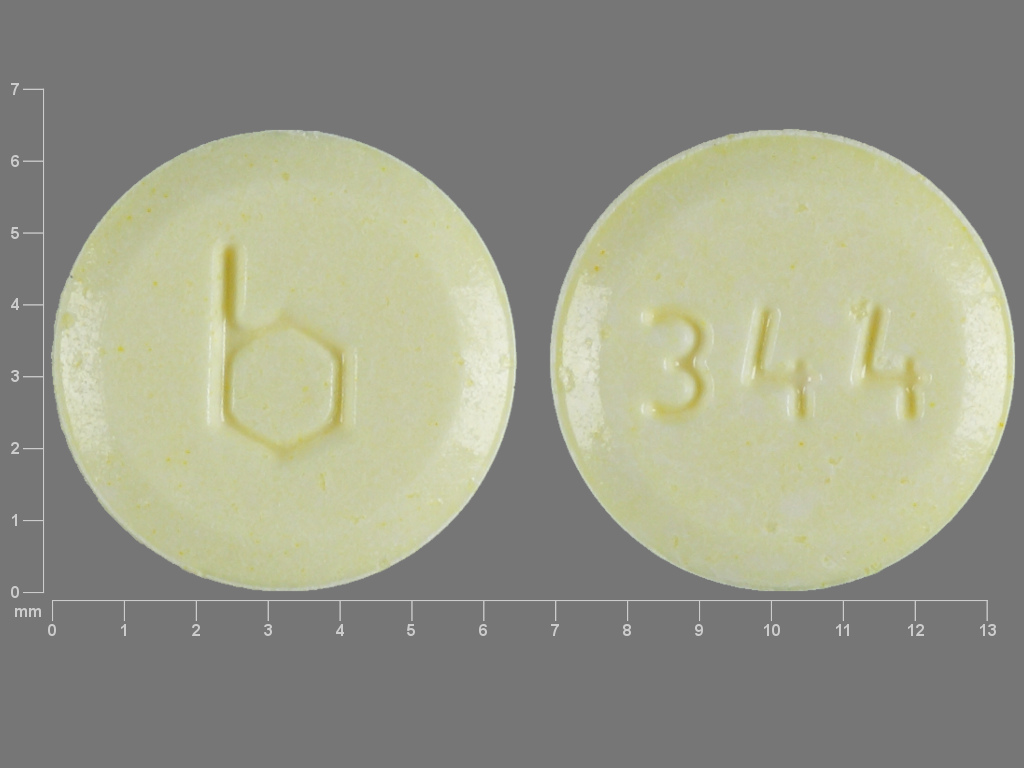
Errin
NORETHINDRONE 0.35 mg;
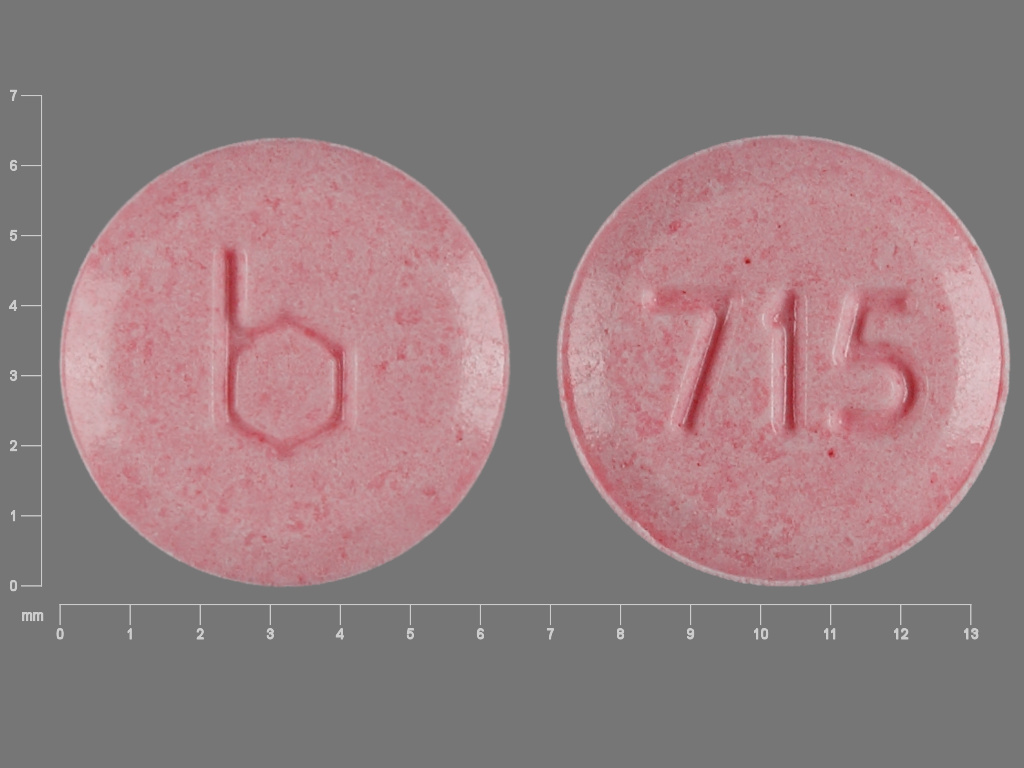
Camila
NORETHINDRONE 0.35 mg;

{12 (Ethinyl Estradiol/Norethindrone/9 (Ethinyl Estradiol/Norethindrone/7 (Inert Ingredients
0.035 MG/0.5 MG/0.035 MG/1 MG/1 MG
NDC: 00555-9066-67
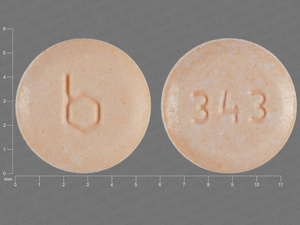
{12 (Ethinyl Estradiol/Norethindrone/9 (Ethinyl Estradiol/Norethindrone/7 (Inert Ingredients
0.035 MG/0.5 MG/0.035 MG/1 MG/1 MG
NDC: 00555-9066-67
What is an imprint code?
An imprint code is a combination of letters, numbers, or symbols that appears on a pill, tablet, or capsule. The FDA requires all approved drug products to have a unique imprint so that they can be identified in case of accidental ingestion or medication confusion. The imprint B is the code found on the pill you are looking up.
⚠️ Disclaimer: Pill identification is for educational purposes only. Always confirm identification with a licensed pharmacist. Read full disclaimer.