Pill With Imprint N
This page shows all pills in our database with the imprint code N. Imprint codes are stamped or printed on pill tablets and capsules and are used to uniquely identify medications. Data is sourced from the FDA NDC Directory and DailyMed.
Found 42 pills with imprint N

Donnatal
PHENOBARBITAL 16.2 mg;HYOSCYAMINE SULFATE 0.1037 mg;ATROPINE SULFATE 0.0194 mg;SCOPOLAMINE HYDROBROMIDE 0.0065 mg;

Mexiletine Hydrochloride
MEXILETINE HYDROCHLORIDE 250 mg;
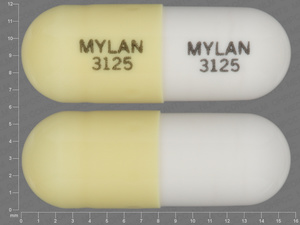
Unknown Pill
NDC: 00904-1261-61

Microencapsulated Potassium Chloride
10 MEQ
NDC: 62037-0710-01
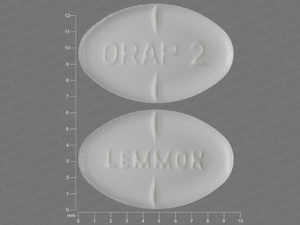
Pimozide
2 MG
NDC: 57844-0187-01
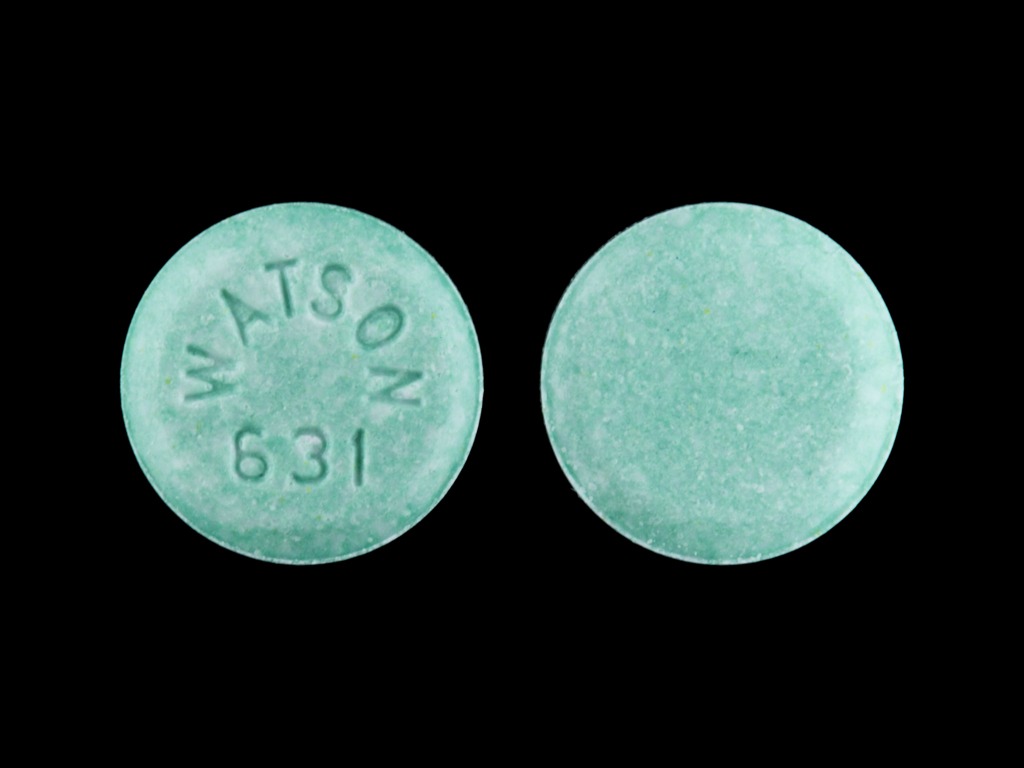
Microgestin Fe
NORETHINDRONE ACETATE 1.5 mg;ETHINYL ESTRADIOL 0.03 mg;
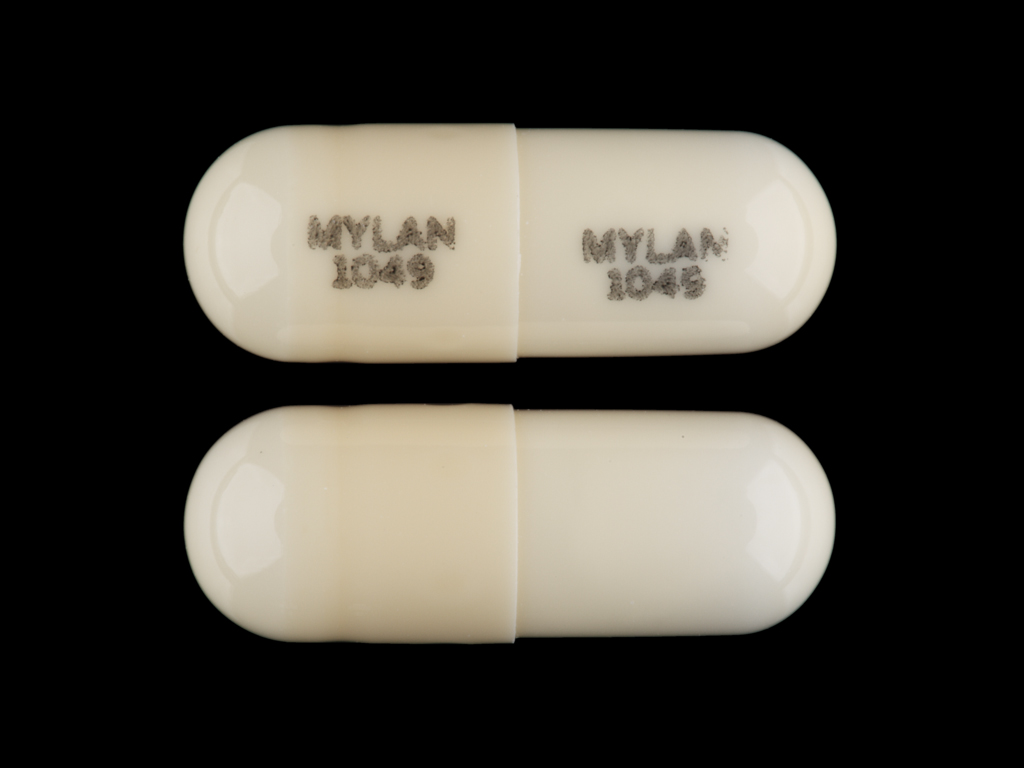
Doxepin Hydrochloride
DOXEPIN HYDROCHLORIDE 10 mg;
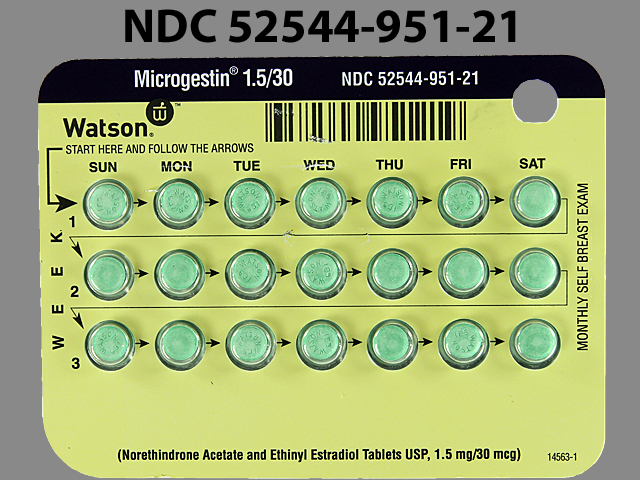
Microgestin
NORETHINDRONE ACETATE 1.5 mg;ETHINYL ESTRADIOL 0.03 mg;
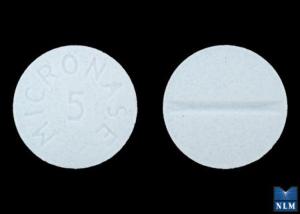
Micronase
glyburide 5 mg
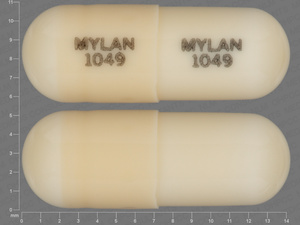
Doxepin Hydrochloride
10 MG
NDC: 51079-0436-20
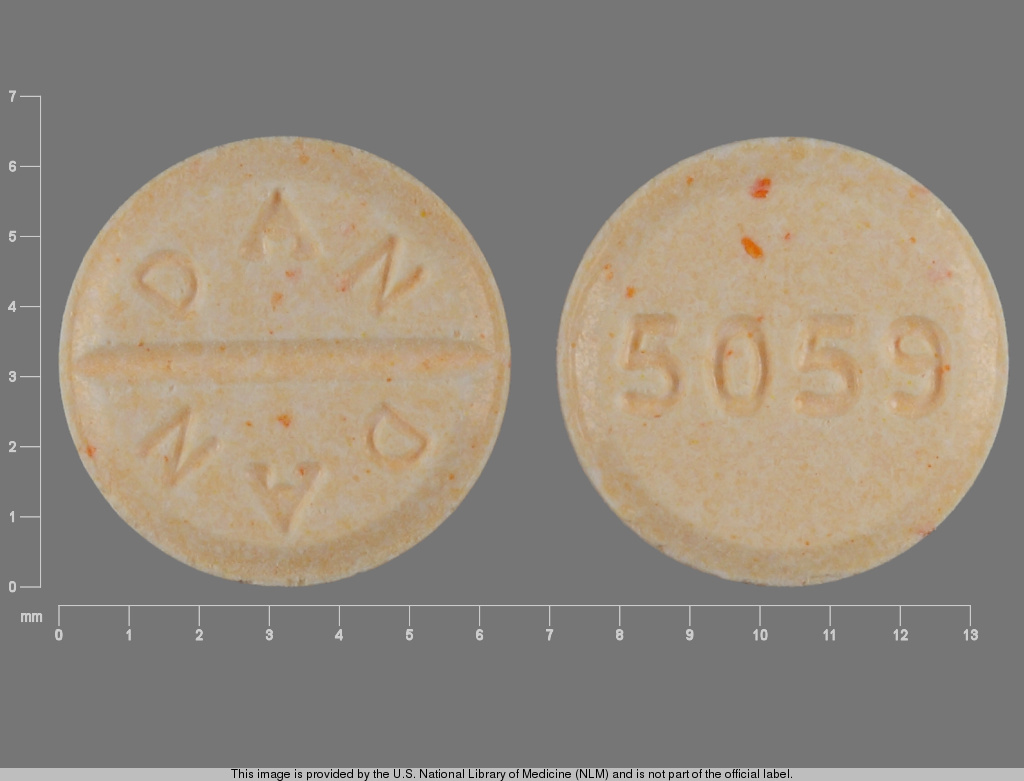
MILLIPRED
PREDNISOLONE 5 mg;
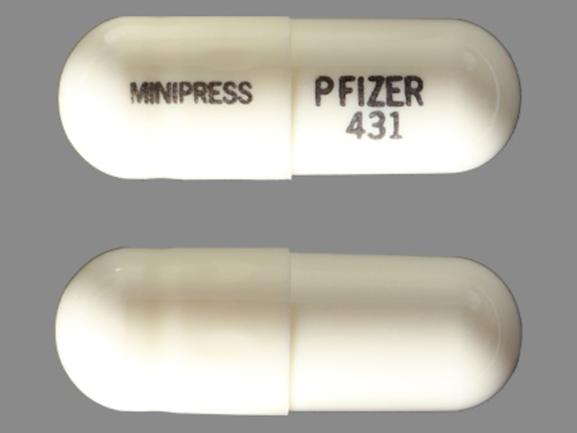
Minipress
PRAZOSIN HYDROCHLORIDE 1 mg;
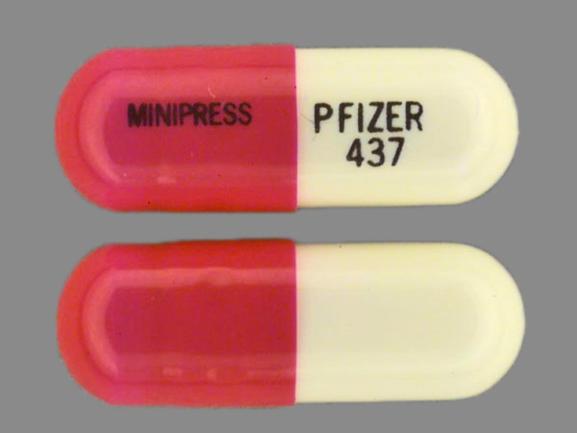
Minipress
PRAZOSIN HYDROCHLORIDE 2 mg;
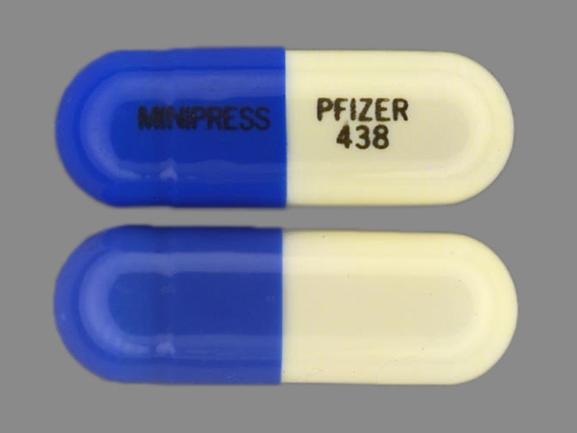
Minipress
PRAZOSIN HYDROCHLORIDE 5 mg;

{21 (Desogestrel/Ethinyl Estradiol/5 (Ethinyl Estradiol/2 (Inert Ingredients
0.15 MG/0.02 MG/0.01 MG/1 MG
NDC: 52544-0940-28
{21 (Ethinyl Estradiol/Levonorgestrel/7 (Inert Ingredients
0.03 MG/0.15 MG/1 MG
NDC: 51285-0091-58

{21 (Ethinyl Estradiol/Norethindrone/7 (Inert Ingredients
0.035 MG/0.4 MG/1 MG
NDC: 52544-0953-28

Unknown Pill
NDC: 65162-0347-58
{7 (Desogestrel/Ethinyl Estradiol/7 (Desogestrel/Ethinyl Estradiol/7 (Desogestrel/Ethinyl Estradiol/7 (Inert Ingredients
0.1 MG/0.025 MG/0.125 MG/0.025 MG/0.15 MG/0.025 MG/1 MG
NDC: 52544-0959-28
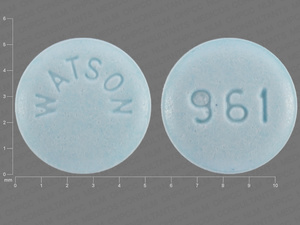
Acebutolol Hydrochloride
ACEBUTOLOL HYDROCHLORIDE 400 mg;
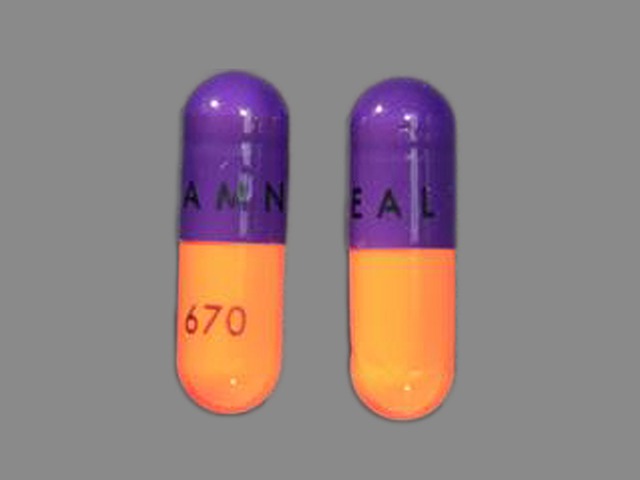
Acetaminophen/Hydrocodone Bitartrate
325 MG/5 MG
NDC: 52544-0913-01
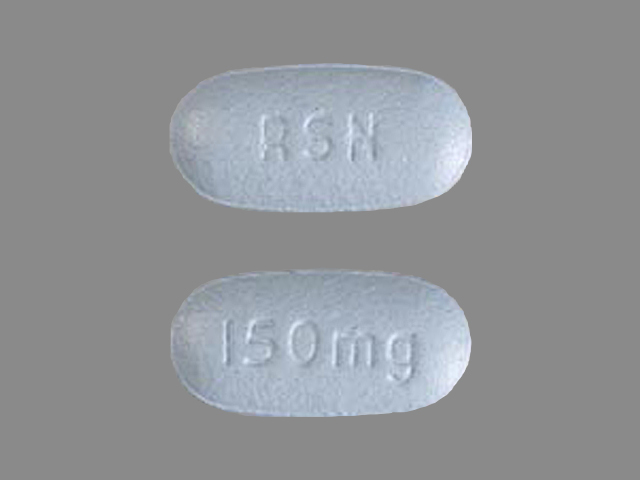
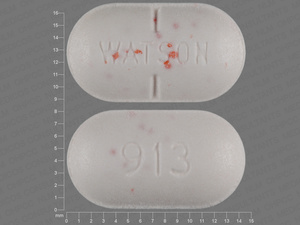
ACTONEL
RISEDRONATE SODIUM 150 mg;
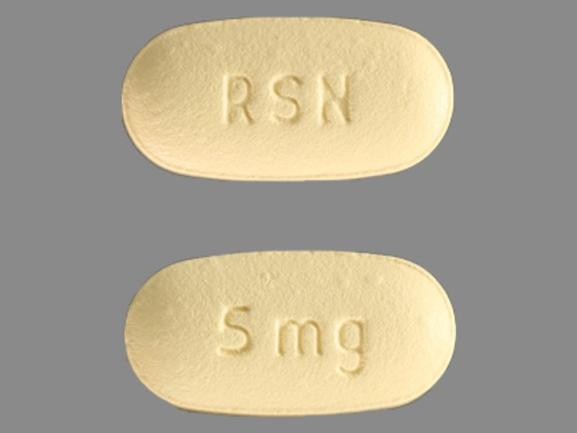
Actonel
RISEDRONATE SODIUM HEMI-PENTAHYDRATE 4.3 mg;RISEDRONATE SODIUM MONOHYDRATE 0.7 mg;
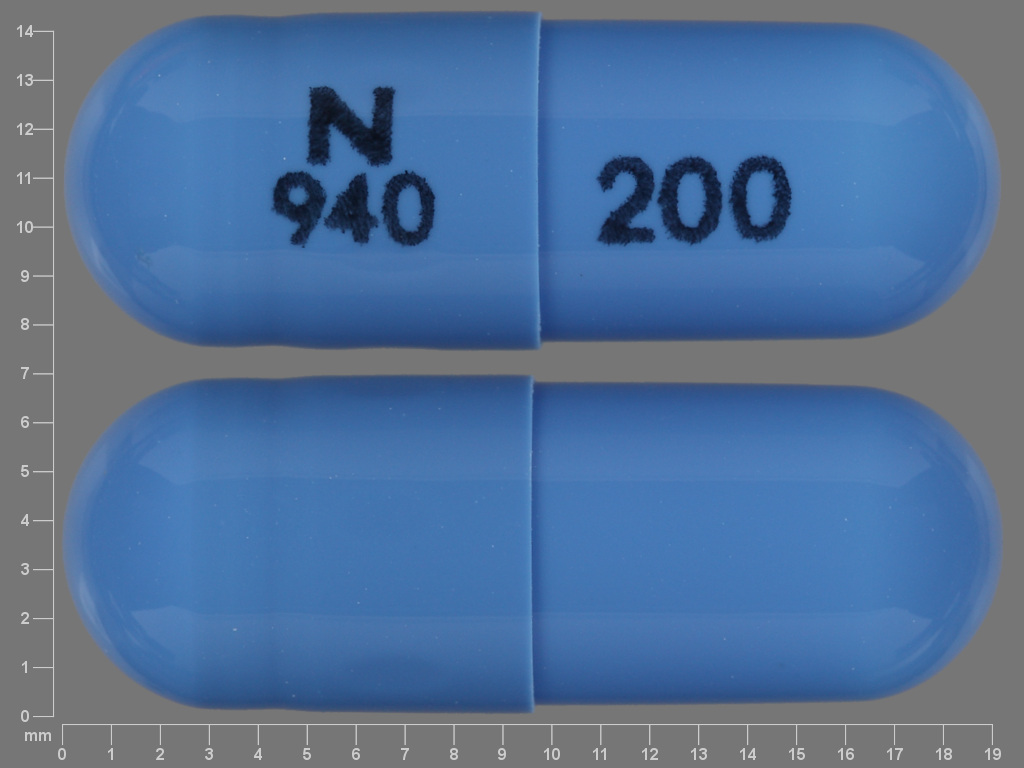
Acyclovir
ACYCLOVIR 200 mg;

Acyclovir
ACYCLOVIR 400 mg;
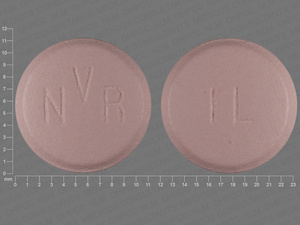
aliskiren
150 MG
NDC: 00078-0485-15
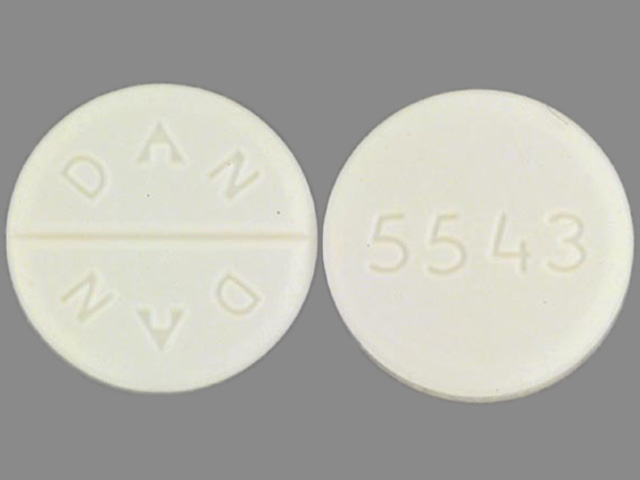
Allopurinol
ALLOPURINOL 100 mg;

Alprazolam
ALPRAZOLAM 1 mg;

Amitriptyline Hydrochloride/Perphenazine
10 MG/2 MG
NDC: 00378-0330-01

Amlodipine besylate/atorvastatin calcium
AMLODIPINE BESYLATE 5 mg;ATORVASTATIN CALCIUM TRIHYDRATE 20 mg;
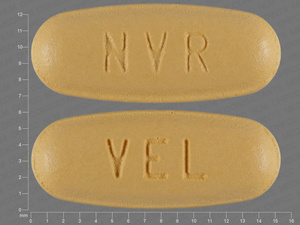
Amlodipine/Hydrochlorothiazide/valsartan
5 MG/25 MG/160 MG
NDC: 00078-0560-15
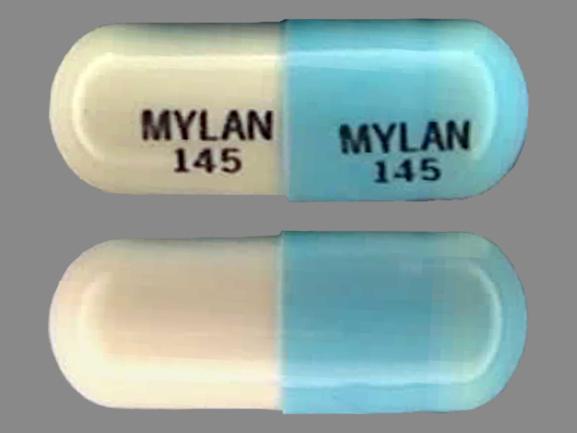
doxycycline hyclate
DOXYCYCLINE HYCLATE 50 mg;

Amylases/Endopeptidases/Lipase
60000 UNT/38000 UNT/12000 UNT
NDC: 00032-1212-01
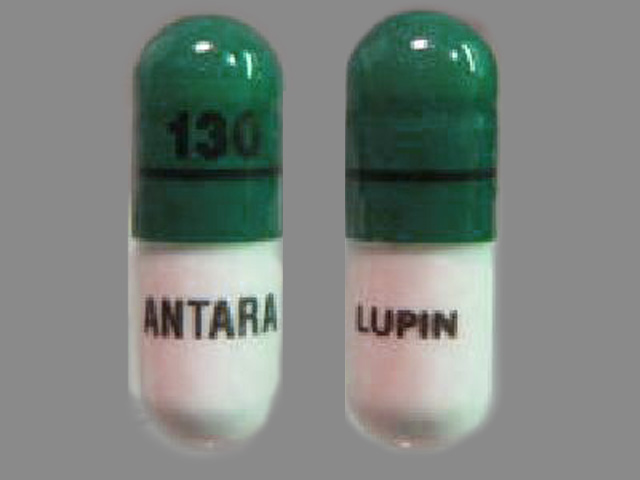
ANTARA
FENOFIBRATE 130 mg;
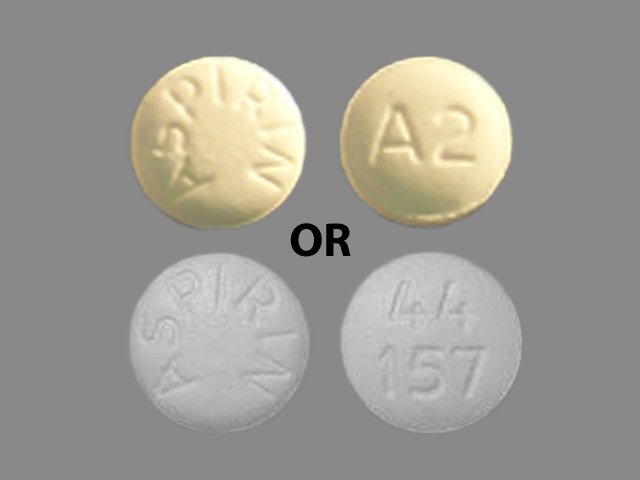
Aspirin
ASPIRIN 325 mg;

Aspirin
ASPIRIN 325 mg;

thyroid (USP)
48.75 MG
NDC: 64727-3302-01

Benicar HCT
OLMESARTAN MEDOXOMIL 20 mg;HYDROCHLOROTHIAZIDE 12.5 mg;

Budesonide
BUDESONIDE 3 mg;
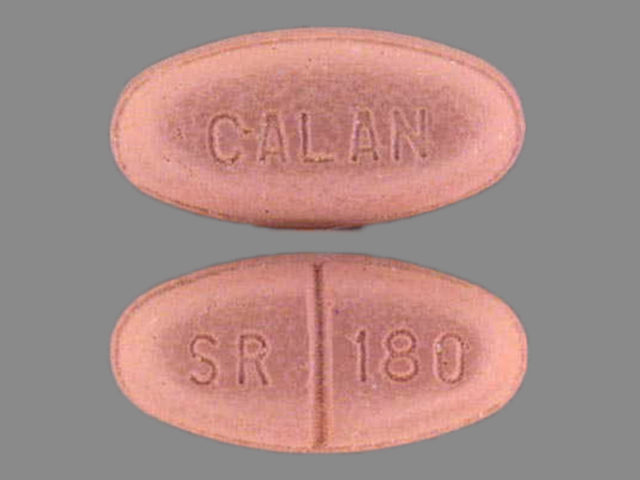
CALAN SR
VERAPAMIL HYDROCHLORIDE 180 mg;
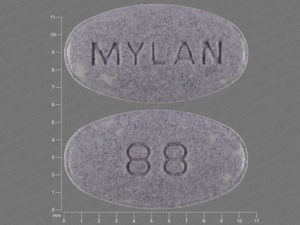
Carbidopa/Levodopa
25 MG/100 MG
NDC: 00378-0088-01

thyroid (USP)
65 MG
NDC: 64727-3300-01
What is an imprint code?
An imprint code is a combination of letters, numbers, or symbols that appears on a pill, tablet, or capsule. The FDA requires all approved drug products to have a unique imprint so that they can be identified in case of accidental ingestion or medication confusion. The imprint N is the code found on the pill you are looking up.
⚠️ Disclaimer: Pill identification is for educational purposes only. Always confirm identification with a licensed pharmacist. Read full disclaimer.